Research Interests
Huang Laboratory Research Overview
The major focus of Dr. Jiaoti Huang's laboratory is the study of prostate cancer. We are interested in different aspects of prostate cancer, including the contribution of cellular heterogeneity to hormone therapy resistance and disease progression, metabolic mechanisms involved in carcinogenesis and cell survival, as well as novel therapeutic approaches for the treatment of castration-resistant prostate cancer.
Our studies are translational in nature and the findings are closely related to human disease. One major strength of our laboratory is the variety of expertise we have in molecular and cellular biology, biochemistry, animal models, histology and immunohistochemistry. Another strength is the large amount of tissue resources available to us. We have access to thousands of cases of archival prostate cancer with long-term follow-up. These include formalin-fixed, paraffin-embedded tissue blocks and frozen tissues. In addition, our lab has pioneered a tissue procurement procedure for fresh prostate tissue from prostatectomy specimens. We routinely process fresh cancer and benign prostate tissues into single cell suspension which has been used in a number of highly impactful publications.
The lab has several major areas of specialization, which include:
Neuroendocrine (NE) cells in prostate cancer.
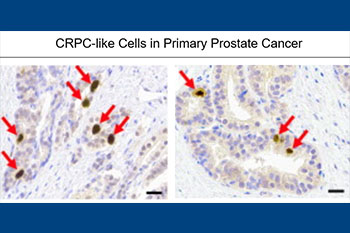
Our lab has had a long-standing interest in NE cells, which comprise a minor component of epithelial cells in benign prostate and adenocarcinoma. These cells lack androgen receptor and are independent of androgen. As a result, they survive and become enriched after hormonal therapy, contributing to the development of castration-resistance. Furthermore, 17-20% of hormonally-treated tumors recur as the highly lethal small cell neuroendocrine carcinoma consisting entirely of NE cells. Therefore, there is an urgent need to define molecular markers for NE cells that can serve as useful therapeutic targets for patients who have exhausted currently used therapies. Using both fresh human prostate cancer tissue, as well as archival formalin-fixed paraffin-embedded tissue specimens, our lab discovers and validates novel molecular markers for this important therapy-resistant tumor cell population. Our lab was the first to report that NE cells in prostate cancer express CXCR2 and GPC3, which has led to the development of novel therapeutic agents as well as ongoing clinical trials.
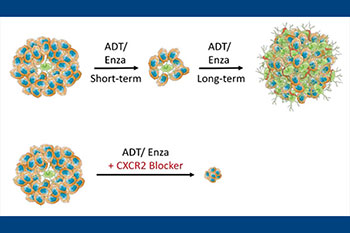
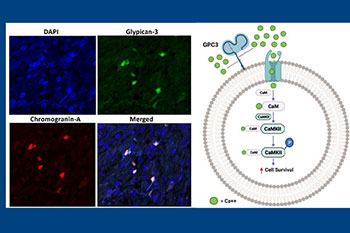
Metabolic mechanisms of disease progression
It is well-accepted that cancer cells display altered metabolism compared to normal cells and that many of these pathways appear critical to their survival. In many cancers, the “Warburg Effect” is the most common observation, characterized by high glucose uptake and increased lactate production to maintain cellular energy needs. However, in most stages of prostate cancer, this effect is not observed and the underlying metabolic anomalies have yet to be discovered. Our lab is interested in defining the major metabolic pathways in advanced prostate cancer as well as discovering ways to target these pathways. Using both primary cells and cell lines engineered to represent various stages of disease, our lab: 1) Identifies novel pathways involved in resistance to hormonal therapy and disease progression, 2) Identifies the major enzymatic drivers of the altered pathways observed, and 3) Develops novel approaches to inhibit the enzymes identified to be involved in disease progression to ultimately suppress prostate cancer growth. This approach led to the discovery that prostate cancer cells become “addicted” to glutamine as a means of surviving hormonal therapy, and that this effect is due to an isoform switch in the major enzyme involved in the catabolism of glutamine (GLS1). Inhibition of the more potent isoform, Glutaminase C, has been shown to result in strong suppression of prostate cancer growth and this work has led to ongoing drug-development to improve the standard of care for patients with castration-resistant prostate cancer.
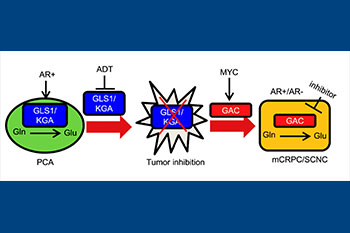
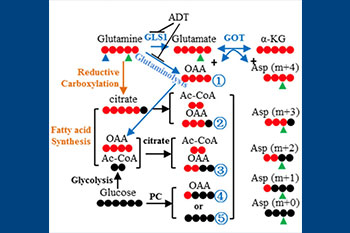
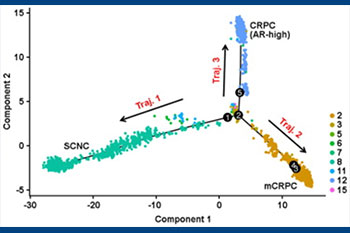
Cellular heterogeneity in prostate cancer
Prostate Cancer demonstrates significant inter- and intra-tumoral heterogeneity which is a major mechanism for resistance to commonly used therapies. By using the latest in sequencing technologies, our lab focuses on the identification of subpopulations of tumor cells responsible for disease progression as well as identifying evolutionary relationships between cell populations to better describe how the disease progresses over time. This led to the discovery that there exists a minor population of cells in primary prostate cancer that are inherently resistant to hormonal therapy and contribute to therapy-failure. Furthermore, we have discovered novel markers and signatures to classify these cells which may help predict the responsiveness to hormonal therapy and survival.
Lab Members

Xue Jiang, PhD
Dr. Jiang is a postdoctoral associate in the Duke Pathology Department. She completed her Ph.D at Kunming institute of Zoology, Chinese Academy of Sciences in China. Her research interests are the pathogenic mechanism and biomarkers of prostate cancer. She is currently working on the role of CXCR1 in progression of prostate cancer under the direction of Dr. Jiaoti Huang.

Chen Jin
Chen Jin is a visiting student in the Duke Pathology Department. He received his master's degree in Urology from Anhui Medical University in China. Currently, he is conducting research on the treatment of advanced prostate cancer by targeting tumor immune microenvironment under the guidance of Jiaoti Huang, MD, PhD.

Hongyang Qian, MD
Dr. Qian is a research scholar in the Duke Pathology Department. He completed his MD at Shanghai Jiao Tong University School of Medicine. His research of interests include pathogenic mechanism of prostate cancer tumorigenesis and Raman Spectroscopy based analysis of cancer. He currently is working on the pathogenic mechanisms of neuroendocrine prostate cancer under the direction of Jiaoti Huang, MD, PhD. .

Cheng Su, MD
Dr. Su is a research scholar in the Duke Pathology Department. His research is centered on the single-cell multi-omics analysis of urogenital tumors. Currently, he is working on the heterogeneity of neuroendocrine cells in prostate cancer at single-cell resolution.

Jianpeng Yu, MD, PhD
Jianpeng Yu is currently a visiting research scholar at the Duke Pathology Department. He received his MD and PhD degrees from the Tianjin Medical University in China. His research interests are clinicopathological characteristics and mechanisms of castration-resistant prostate cancer progression. He currently is working on metabolic reprogramming in prostate cancer under the direction of Jiaoti Huang, MD, PhD

Fang Zhang, PhD
Dr. Zhang is a postdoctoral associate in Duke Pathology. She received her PhD degree from the School of Life Sciences, Fudan University in China, where she explored therapeutic targets in pituitary neuroendocrine tumors (PitNET) based on a proteogenomics strategy. She currently is performing metabolic studies and omics analyses to discover more effective therapies for advanced and therapy-resistant prostate cancers under the direction of Jiaoti Huang, MD, PhD.
Publications
Please see Pub Med for current listings.
Book Sections
- Koo, J, Filson, CP, Huang, J, and Pantuck, AJ."Collecting Duct Carcinoma and Renal Medullary Carcinoma."In The Kidney: A Comprehensive Guide to Pathologic Diagnosis and Management, edited by DE Hansel, CJ Kane, GP Paner, and S Chang,109-123. 2016. (Chapter)
- Brick, A, Niu, J, Huang, J, and Oh, WK. "Possibility for Platinum Chemotherapy Treatment for HRPC Patients?." In Drug Management of Prostate Cancer, edited by WD Figg, CH Chau, and EJ Small,153-161. NJ: Humana Press, 2010. (Chapter)
- Wu, C, Chen, G, Na, Y, Xin, D, Yao, JL, di Xant' Agnese, PA, and Huang, J. "The function of Interleukin-8 in prostate cancer." In Progress in Chemokine Research, edited by WP Links,149-156. New York: Nova Science Publishers, 2007. (Chapter
Conference Papers
- Samaratunga, H, Montrironi, R, True, L, Epstein, JI, Griffiths, DF, Humphrey, PA, van der Kwast, T, Wheeler, TM, Srigley, JR, Delahunt, B, Egevad, L, and ISUP Prostate Cancer Group,."Working group 1: specimen handling."International Society of Urological Pathology (ISUP) Consensus Conference on Handling and Staging of Radical Prostatectomy Specimens. Nature Publishing Group, 2011
- Huang, J, Wu, C, Yao, JL, and di Sant'Agnese, PA. "Neuroendocrine differentiation in prostate cancer." Bologna, Italy. Medimond S.r.l., 2005.
Journal Articles
- Duvnjak, P, Schulman, AA, Holtz, JN, Huang, J, Polascik, TJ, and Gupta, RT. "Multiparametric Prostate MR Imaging: Impact on Clinical Staging and Decision Making." Radiologic clinics of North America 56, no. 2 (March 2018): 239-250
- Holtz, JN, Silverman, RK, Tay, KJ, Browning, JT, Huang, J, Polascik, TJ, and Gupta, RT. "New prostate cancer prognostic grade group (PGG): Can multiparametric MRI (mpMRI) accurately separate patients with low-, intermediate-, and high-grade cancer?." Abdominal radiology (New York) 43, no. 3 (March 2018): 702-712.
- Okhunov, Z, Yoon, R, Lusch, A, Spradling, K, Suarez, M, Kaler, KS, Patel, R, Hwang, C, Osann, KE, Huang, J, Lee, TK, and Landman, J. "Evaluation and Comparison of Contemporary Energy-based Surgical Vessel Sealing Devices." Journal of endourology (February 20, 2018)
- Nagarajan, MB, Raman, SS, Lo, P, Lin, W-C, Khoshnoodi, P, Sayre, JW, Ramakrishna, B, Ahuja, P, Huang, J, Margolis, DJA, Lu, DSK, Reiter, RE, Goldin, JG, Brown, MS, and Enzmann, DR. "Building a high-resolution T2-weighted MR-based probabilistic model of tumor occurrence in the prostate." Abdominal radiology (New York) (February 19, 2018)
- Nassiri, N, Chang, E, Lieu, P, Priester, AM, Margolis, DJA, Huang, J, Reiter, RE, Dorey, FJ, Marks, LS, and Natarajan, S. "Focal Therapy Eligibility Determined by Magnetic Resonance Imaging/Ultrasound Fusion Biopsy." The Journal of urology 199, no. 2 (February 2018): 453-458
- Chang, E, Jones, TA, Natarajan, S, Sharma, D, Simopoulos, D, Margolis, DJ, Huang, J, Dorey, FJ, and Marks, LS. "Value of Tracking Biopsy in Men Undergoing Active Surveillance of Prostate Cancer." The Journal of urology 199, no. 1 (January 2018): 98-105
- Felker, ER, Raman, SS, Margolis, DJ, Lu, DSK, Shaheen, N, Natarajan, S, Sharma, D, Huang, J, Dorey, F, and Marks, LS. "Risk Stratification Among Men With Prostate Imaging Reporting and Data System version 2 Category 3 Transition Zone Lesions: Is Biopsy Always Necessary?." AJR. American journal of roentgenology 209, no. 6 (December 2017): 1272-1277
- Chen, W-Y, Tsai, Y-C, Siu, MK, Yeh, H-L, Chen, C-L, Yin, JJ, Huang, J, and Liu, Y-N. "Inhibition of the androgen receptor induces a novel tumor promoter, ZBTB46, for prostate cancer metastasis." Oncogene 36, no. 45 (November 2017): 6213-6224
- Alle, N, Tan, N, Huss, J, Huang, J, Pantuck, A, and Raman, SS. "Percutaneous image-guided core biopsy of solid renal masses: analysis of safety, efficacy, pathologic interpretation, and clinical significance." Abdominal radiology (New York) (October 27, 2017)
Former Graduate Students and Postdoctoral Trainees
Many outstanding visiting scholars, undergraduate students, and Graduate Program in Pathobiology and Translational Biosciences rotation students have also participated in the lab's research activities over the many years.
Former Graduate Students:
- William Butler, PhD (2018-2023)
- Yanjing Li, PhD (2013-2017)
Former Postdoctoral Trainees:
- Spencer Hauck, PhD (2020-2024)
- Lingfan Xu, MD (2016-2021)
Former Medical Students
- Jerry Fu (2021-2023)
- David Moon (2021-2024)